Researchers Seek Funding Partners for 21st Century Chain Link Armor
A team of researchers at Northwestern University recently announced a breakthrough nanomaterial—a two-dimensional (2D) polymer that mechanically interlocks like chainmail—that could lead to stronger, more flexible body armor. But government funding from the National Science Foundation (NSF) and the Defense Advanced Research Projects Agency has ended, leaving the scientists searching for new funding.
The NSF announced the breakthrough in March, but when SIGNAL Media contacted the researchers the following month, the NSF’s support had ended. “We have no funding for this project as of today—not for lack of trying—but we have grants that we have submitted and hopefully will be successful. I think they’re extremely compelling, given what we’ve discovered,” said William Dichtel, Northwestern University researcher and professor.
The Northwestern team published the results of their work in the journal Science, along with researchers from Cornell University, who used an advanced imaging technique to show the microscopic details of the new material, the first mechanically interlocking 2D polymer.
The NSF press release explains that the creation process involves coaxing polymers to form mechanical bonds, a feat that has challenged researchers for years. The Northwestern team created a novel process to form the bonds by arranging ordered crystalline structures of polymer molecules and then causing the crystals to react with another molecule to create bonds inside the crystal’s molecules. The resulting crystals comprise layers and layers of 2D interlocked polymer sheets.
Doctoral candidate Madison Bardot, the first author for the research paper, had the initial “aha!” moment that led to the innovative breakthrough. “I absolutely love crystals, so one day I was just reading through the literature, and I saw what they call hydrogen-bonded frameworks. Basically, molecules were like each other in crystals. And I thought, wow, what if you could take molecules, make them sit next to each other in crystals, and then react the crystal,” Bardot explained to SIGNAL Media. “At first, to a chemist, that sounds a little bit crazy because normally, once you’ve made a crystal, you’re very excited, and you don’t want to destroy that crystal, but it’s actually turned out to be a really great platform for us.”
Dichtel also credits Bardot for being modest in her description of the breakthrough. “What happened here is the best aspect of science. Madison is a young researcher doing her Ph.D. and thinking about the possibilities of reactions that have never happened before,” he reported. “She found a paper and had this idea that a more experienced person might have dismissed as either difficult or naive, and she decided to try it anyway, and it works spectacularly well.”
The research paper describes a highly efficient and scalable polymerization process that could potentially yield high volumes of the material at mass scale, scalability being another major achievement. In addition to being the first 2D mechanically interlocked polymer, it also contains 100 trillion mechanical bonds per 1 square centimeter—the highest density of mechanical bonds ever achieved in a material, according to the NSF press release. “We made a completely new polymer structure,” Dichtel said in the release. “It’s similar to chainmail in that it cannot easily rip because each of the mechanical bonds has a bit of freedom to slide around. If you pull it, it can dissipate the applied force in multiple directions.”
The 2016 Nobel Prize in Chemistry was awarded to researchers who formed a chain of ring-shaped molecules, which could be the beginning of molecular machines, Dichtel noted. “But those are all kind of fancy and exotic molecules that people were making in very tiny amounts and studying them at a very fundamental level,” he said.
It has long been speculated that polymers with lots of interlocked rings would have very different mechanical properties than traditional polymers that are either long entangled chains or infinite cross-linked networks, he added. “So, it’s been thought that these mechanical bonds would be very, very interesting to put into polymers, but there just wasn’t any way to do it on any practical level prior to the work that Madison and I and our coauthors reported recently.”
Dichtel explained to SIGNAL Media that while the material is flexible, it hardens when pressure is applied.
“The most notable thing is that these materials harden, which is kind of expected from a structure that is interlocked like this. When you start pushing on them, they’ve got a lot of give to them, because the mechanical bonds can just sort of move very easily with respect to each other,” he said. “But at some point, if you keep pushing on them, you reach the limits of the interlock structures, and then they become much, much harder, like a traditional polymer. That combination of properties is definitely of interest for really tough materials like armor.”
Indeed, collaborating researchers from Duke University added the Bardot polymer to ULTEM, a strong, protective material in the same family as Kevlar. The researchers developed a composite material of 97.5% ULTEM fiber and just 2.5% chainmail polymer, which significantly boosted ULTEM’s toughness. “Almost every property we have measured has been exceptional in some way,” Dichtel declared.
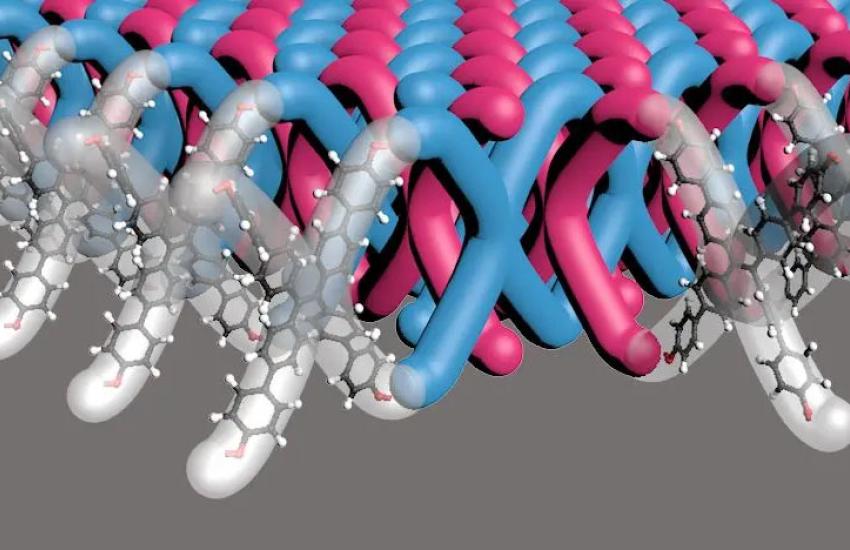
That breakthrough demonstrates another unique property of Bardot’s polymer: its dissolvability, which allows it to be easily integrated with other materials. “The hard thing about working with large polymers is usually the larger the polymer you get, the harder it is to incorporate into other things. I always like to use the example of, like, a piece of tire. You’re not going to be able to dissolve that and then incorporate it into other things. But in our material, what’s really nice is we are able to actually put these into solvent, and that means that when we’re trying to put them into fibers, or if we wanted to coat them on a surface, we can actually have a way to process the material.”
The next steps, according to the researchers, will be to continue modifying and studying the materials, which may lead to a whole new class of materials. “We are really interested in seeing how broad a class of materials this might be. The paper reports the first one, but there are a number of ways to modify the structure and study more mechanically interlocked polymers that can be adapted to speculative applications. We’re looking at different chemical structures while still maintaining the ability to make large quantities of these materials,” Dichtel said.
He added that it’s interesting to find building blocks that are similar and will allow other mechanical bonds to be formed using Bardot’s unique strategy. “It’s also very interesting to take the existing material and tweak the structure a little bit and see how broadly this idea can be applied with materials offering different properties. We really only scratched the surface of what is possible so far.”
The science is still in the early, basic research phase, but such materials might ultimately be used for purposes other than body armor. “There’s all kinds of areas where you’d want really flexible yet tough materials,” Dichtel said. “We haven’t examined any of these, but you can imagine, like separators in a battery need to have these properties and also have ionic conductivity and things like that.”
New kinds of membranes might also be possible. “These materials have little gaps in each repeat unit—kind of like how water can pass through a woven mesh pretty easily because there’s little gaps between the fibers. So, these may be very interesting for certain kinds of membrane-based separations,” Dichtel offered. “We can speculate about [uses] because this is such a new architecture in polymers that it’s going to take a while for people to figure this out. But, generally, anytime there’s something this new, there will be definitely unique properties that arise.”
Dichtel referred to the research as an “orphaned program” because of the government funding ending, but he added: “I think we turned it into a pretty big success story with this material, so this is something we’re currently seeking funding for.”





Comments